Closed-Loop Systems for Diabetes: Automated Insulin Delivery Guide

| Metric | Manual Pump Therapy | Closed-Loop Systems |
|---|---|---|
| Average Time-in-Range (TIR) | 50-60% | 70-75% |
| Hypoglycemia Risk | Higher (5-7% below 70 mg/dL) | Lower (2-4% below 70 mg/dL) |
| Cognitive Burden | High (Constant calculations) | Reduced (Automated adjustments) |
| HbA1c Impact | Baseline | Typical drop of 0.3-0.5% |
How Closed-Loop Systems Actually Work
At its core, a Closed-Loop System is a feedback loop. It doesn't just pump insulin; it thinks and reacts. To make this happen, the system relies on three specific parts working in perfect harmony: a Continuous Glucose Monitor (CGM), an insulin pump, and a sophisticated algorithm. First, the CGM sensor sits under the skin and checks glucose levels every few minutes. It sends this data via Bluetooth to the algorithm, which acts as the "brain." This brain looks at where your sugar is now and where it's headed. If you're drifting too high, it nudges the pump to deliver more insulin. If you're crashing, it throttles back or shuts off delivery entirely to prevent a severe hypo. This constant conversation happens in the background, allowing you to focus on your life rather than your glucometer.Hybrid vs. Fully Closed-Loop: What's the Difference?
Most of the devices you see today are Hybrid Closed-Loop (HCL) systems. They are called "hybrid" because they aren't totally autonomous. You still have to tell the system when you're eating by entering your carb count-this is called a meal bolus. The system handles the basal (background) insulin, but it can't yet "see" a slice of pizza coming before the glucose actually hits your bloodstream. On the other hand, fully closed-loop systems aim to remove the need for meal announcements. While still largely in the development or early adoption phase, devices like the iLet from Beta Bionics are pushing the needle. Instead of requiring complex insulin-to-carb ratios, the iLet uses an adaptive algorithm that only needs your body weight to get started. It learns your patterns over time and adjusts itself, though it still suggests meal announcements for the best results after eating.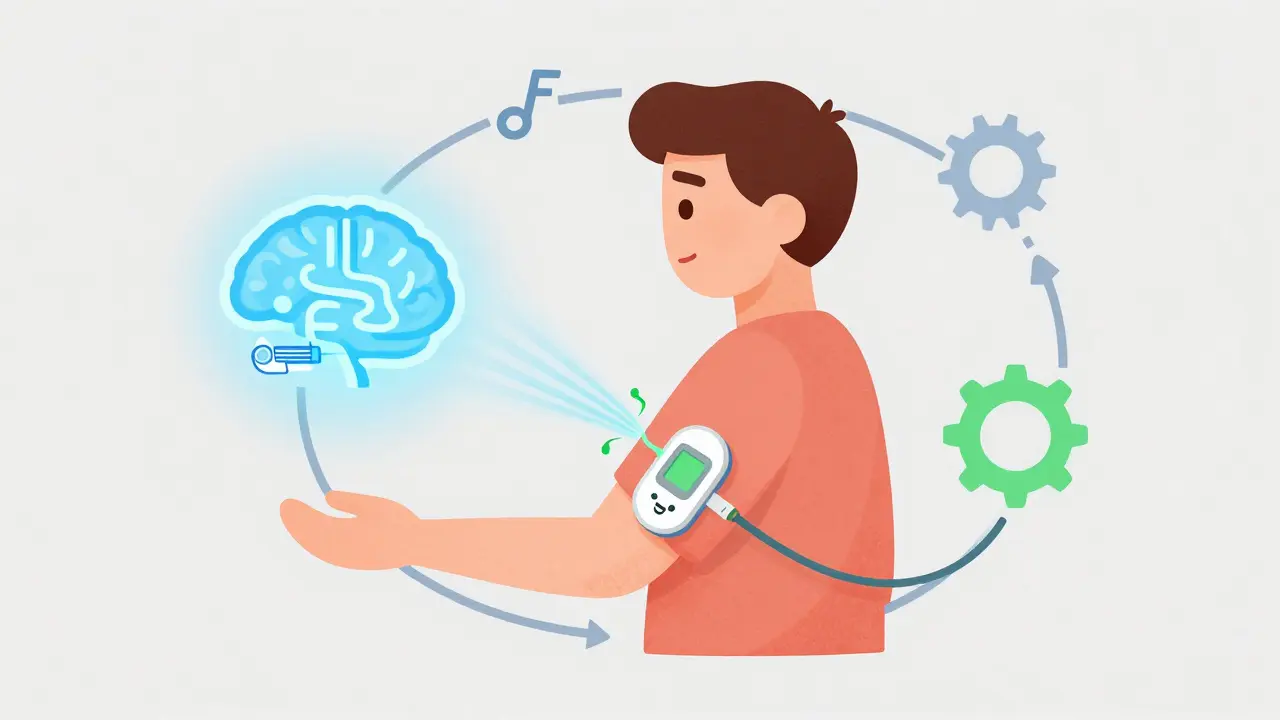
Comparing the Top AID Systems on the Market
Choosing a system usually comes down to how much control you want and whether you prefer a tubed pump or a patch pump. Tandem Diabetes Care offers the t:slim X2 with Control-IQ technology. It's a powerhouse for those who want active correction. Its standout feature is the ability to deliver automatic correction boluses if the algorithm predicts you'll stay above target. However, users have noted a slight lag-about 20 minutes-before the system responds to rapid spikes. If you hate the idea of tubes, the Omnipod 5 by Insulet is the go-to. It's a tubeless patch pump that sticks directly to your skin. It's incredibly discreet and simplifies the hardware side of things, although it historically required more manual meal announcements than its competitors (a point they've worked to improve with recent software updates).Real-World Results: Does the Tech Actually Help?
Clinical data is one thing, but real-world experience tells the full story. In the CLEAR study, participants reported a profound shift in their quality of life. One user described it as "diabetes working around my life rather than my life working around diabetes." For many, the biggest win is sleep. A huge percentage of users on forums like the T1D Exchange report better sleep quality because the system catches overnight lows before they become emergencies. No more waking up drenched in sweat because of a midnight drop. However, it's not a magic wand. There are a few common pitfalls:- Sensor Lag: CGMs measure interstitial fluid, not blood. This means there is a 5-15 minute delay. If your sugar is dropping fast, the pump might still think you're stable for a few minutes.
- Algorithm Fatigue: Some users feel overwhelmed by the constant alerts or the need to override the system during complex activities like high-intensity exercise.
- The DKA Risk: Some data suggests a slightly higher rate of Diabetic Ketoacidosis (DKA) in HCL users. This often happens if a user relies too heavily on the automation and ignores the signs of rising ketones.

Practical Tips for Getting Started
Switching to an automated system isn't as simple as plugging in a toaster. There is a learning curve that usually takes 2 to 4 weeks. If you're making the jump, keep these practical strategies in mind:- Pre-bolus for Success: Since there is a lag in how algorithms respond to meals, try bolusing 15-20 minutes before you eat high-carb meals. This helps the system stay ahead of the spike.
- Use "Exercise Mode": Don't let the system fight your workout. Most AID systems have a temporary target or exercise mode that raises the glucose goal to prevent hypos while you're active.
- Fight Adhesion Issues: Skin irritation is common. Many experienced users swear by using Skin Tac or similar medical adhesives to keep the pump and sensor from peeling off early.
- Calibrate Carefully: If your CGM feels "off," don't just blindly trust the algorithm. Use a finger-stick meter to verify before making a massive correction bolus.
The Road Ahead: What's Next for AID?
We are moving toward an era of "interoperable" systems. This means you won't be locked into one brand's ecosystem. Imagine using a Dexcom sensor with a Tandem pump and a third-party app to analyze the data-all talking to each other seamlessly. Engineers are also looking beyond just glucose. Future algorithms are expected to incorporate stress biomarkers and heart rate data from smartwatches to predict sugar swings before they even start. Within the next five years, most endocrinologists expect fully closed-loop systems-those that require zero one-off inputs for meals-to become the standard of care.Is a closed-loop system safe for children?
Yes, and in fact, adoption is highest among children (about 35% of pump users). These systems provide a safety net that is invaluable for parents, as they can often monitor their child's levels remotely and the system automatically prevents severe hypoglycemia during the night.
Do I still need to count carbs with a hybrid system?
Yes. In hybrid closed-loop systems, you must still announce your meals and enter the estimated carbohydrates. The automation handles the basal rate and minor corrections, but it cannot accurately predict the insulin needed for a meal without your input.
What happens if my CGM sensor fails?
If the sensor fails or loses signal, the system will automatically drop back into "open-loop" mode. This means it reverts to your pre-set basal rates, acting like a traditional insulin pump until the sensor is replaced or the signal returns.
How much do these systems cost?
Costs vary widely. A t:slim X2 pump can retail around $6,500, whereas the Omnipod 5 uses a subscription-like model with pods costing around $320 each (lasting 3 days). Insurance coverage varies, and some patients may face a 20% co-pay depending on their plan.
Can these systems help with my HbA1c?
Yes. Clinical trials show that moving to an HCL system typically reduces HbA1c by 0.3 to 0.5 percentage points compared to standard sensor-augmented pump therapy, largely by increasing the time spent in the target glucose range.
the skin tac mentioned is a total lifesaver for the omnipod 5 especially if you sweat a lot or have oily skin just apply it in a thin layer and let it dry completely before sticking the pod on it makes a huge difference in how long the pods actually stay put
Oh wow!!! Just wow!!! 🙄 I'm sure the insurance companies are just THRILLED to pay for these $6,500 toasters!!! 💸✨ Truly a miracle of modern capitalism!!! 🌈💖
too long
My god, the sheer terror of a midnight crash is a trauma no one talks about! I remember one night where I woke up shaking and I literally thought it was the end of my existence! This technology isn't just a guide, it's a lifeline! I cannot even imagine going back to manual calculations after tasting this kind of freedom!
The notion that these systems provide an "invaluable safety net" for parents is a gross oversimplification of the psychological dependency created by such devices. It merely shifts the anxiety from the child's glucose level to the reliability of the Bluetooth connection, which, as any seasoned user knows, is temperamental at best.
You all need to realize that we are living in a golden age of medical innovation and every single one of you who is struggling right now should look at this as a stepping stone toward a completely cure-oriented future because the momentum of AI and closed-loop integration is moving so fast that we will soon be looking back at the concept of manual insulin dosing as a primitive relic of the past and that is why we must keep pushing our doctors for the latest tech regardless of the initial hurdles!
Typical corporate propaganda pushing these algorithmic shackles on us. They want us dependent on a "brain" owned by a company that probably sells our biometric data to the highest bidder in a shadowy boardroom. This whole "interoperable" dream is just a fancy way of saying they want a backdoor into every single one of your veins. Absolute madness!
Existence is basically just a series of inputs and outputs, and we're just trying to optimize the biological hardware with silicon. If the algorithm is the brain, then we're just the chassis. It's a bleak reflection of where humanity is heading when we outsource our survival to a piece of code.
It is interesting to consider the shift in identity when a machine begins to manage a core bodily function. We move from being active participants in our own survival to becoming monitors of a system. There is a certain philosophical tension between the comfort of safety and the loss of intuitive bodily awareness.
I must insist that every individual who is hesitant to adopt this technology is doing a grave disservice to their own health! It is absolutely imperative that you cease your hesitation and embrace these advancements immediately for the sake of your longevity! Only the weak-willed succumb to fear when a proven scientific solution is presented!
Honestly, if you aren't using the top-tier US-made tech, are you even managing your diabetes? 💅 The American engineering in these systems is light years ahead of anything else. It's honestly embarrassing that some people still settle for less when they could have the gold standard of medical care! 🇺🇸✨
You must be aggressive in your pursuit of better health! Stop making excuses about the learning curve and just do the work! The data proves these systems work, so any refusal to transition is simply a refusal to improve your life! Get it done!